Arch. Endocrinol. Metab. 2021;65(2):185-197
Efficacy of a glucose meter connected to a mobile app on glycemic control and adherence to self-care tasks in patients with T1DM and LADA: a parallel-group, open-label, clinical treatment trial
DOI: 10.20945/2359-3997000000334
ABSTRACT
Objective:
The main aim of the study was to evaluate the patients’ glycemic control and adherence to self-care tasks.
Materials and methods:
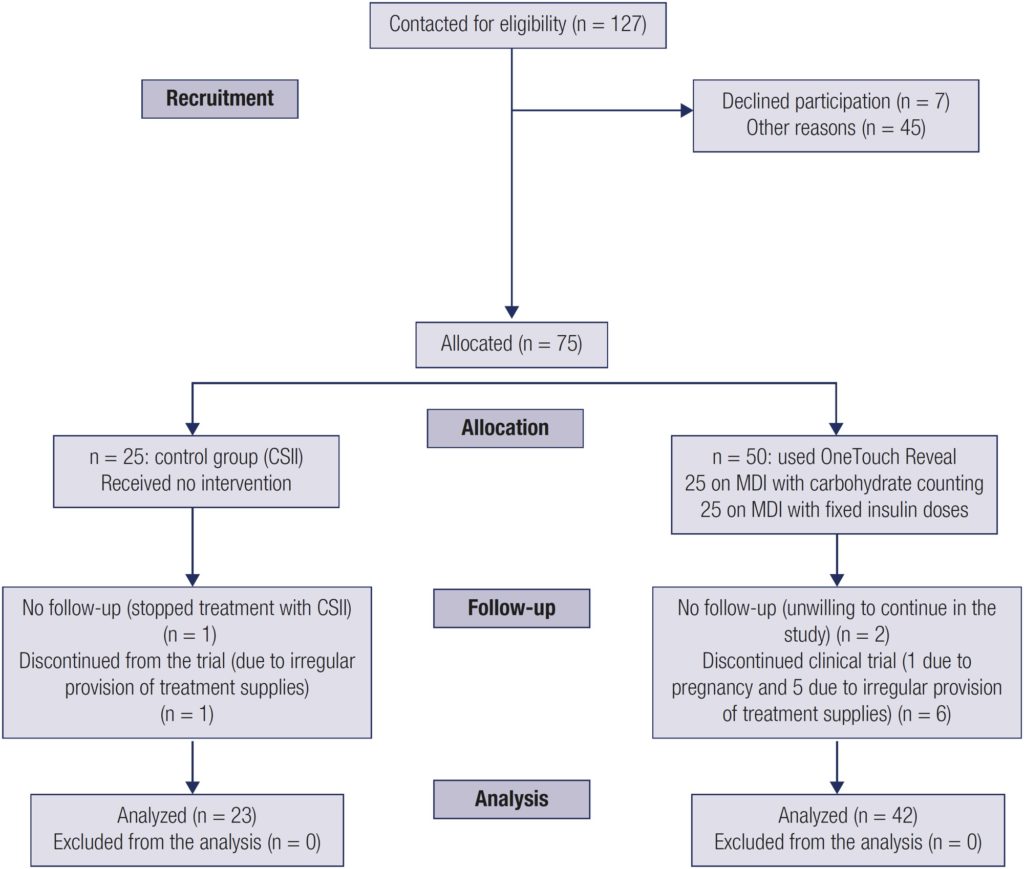
Patients with type 1 diabetes mellitus (T1DM) or latent autoimmune diabetes of the adult (LADA) using a multiple daily injection (MDI) regimen with carbohydrate counting (n = 25, Subgroup B) or fixed insulin dose (n = 25, Subgroup C) were allocated to use the application (app) for 12 weeks. Both subgroups were compared with each other and against a control group (n = 25, Group A) comprising patients with T1DM or LADA treated with continuous subcutaneous insulin infusion (CSII) in a parallel-group, open-label, clinical treatment trial. All patients had glycated hemoglobin (A1C) levels measured and were asked to fill out the Diabetes Self-Management Profile (DSMP) questionnaire at study start and end. The patients were instructed to measure capillary glucose six times daily in study weeks 4, 8, and 12.
Results:
Mean A1C levels decreased 0.725% in Subgroup C in intragroup analysis (p = 0.0063), and had a mean variation of 0.834% compared with Group A (p = 0.003). Mean DSMP scores increased 5.77 points in Subgroup B in intragroup analysis (p = 0.0004) and increased by a mean of 6.815 points in relation to Group A (p = 0.002).
Conclusion:
OneTouch Reveal improved both A1C levels and DSMP scores in patients with T1DM or LADA compared with standard treatment (CSII).