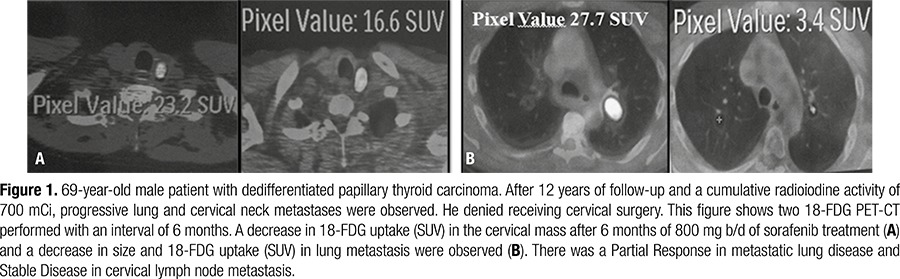
Arq Bras Endocrinol Metab 2014;58(1):37-41
Response to sorafenib treatment in advanced metastatic thyroid cancer
DOI: 10.1590/0004-2730000002839
Objective
: To investigate the efficacy of sorafenib in progressive radioiodine resistant metastatic thyroid carcinoma.
Subjects and methods
: Off-label observational study. Sorafenib 400 mg twice daily was evaluated. Therapy duration was 12 ± 3 months (range 6-16 months).
Results
: Eight patients were included (seven papillary, one insular variant). The eight patients meeting study criteria received sorafenib 400 mg orally twice a day until disease progression or unacceptable toxicity developed. One patient showed a partial response with tumor regression of -35%, six months after the beginning of the treatment; five patients exhibited stable disease and two patients had progressive disease and died. Thyroglobulin decreased within 4 weeks in all patients by 50% ± 23%.
Adverse events
: one patient had heart failure, and recovered after sorafenib withdrawal. However, she died five months later of sudden death.
Conclusion
: These data suggest a possible role for sorafenib in the treatment of progressive metastatic DTC. Adverse event are usually manageable, but severe ones may appear and these patients should be strictly controlled.
Keywords: cancer; Metastasis; sorafenib; thyroid